What is the mechanism of an oil refinery, which produces petroleum, a material widely used as fuel and a raw material for synthetic fibers?

Even with the growing focus on renewable energy, more than 100 million
How an Oil Refinery Works - by Brian Potter
https://www.construction-physics.com/p/how-an-oil-refinery-works
Petroleum is not only an excellent fuel, but also an excellent raw material, as evidenced by the fact that 90% of chemical raw materials are made from petroleum or natural gas. All plastics are made from chemicals extracted from petroleum or natural gas, and petrochemical products range from lubricants and paints to plywood, synthetic fibers, and fertilizers.
The ability to consume vast quantities of oil is thanks to oil refineries. When oil is extracted from the earth, it is crude oil, a complex mixture of thousands of different chemical substances. Oil refineries play the role of processing this mixture and transforming it into usable chemical substances.
Considering global oil consumption, oil refineries are among the world's largest industrial facilities. Large-scale oil refineries occupy millions of square meters of land and cost hundreds of billions of yen to construct.

Petroleum is essentially a liquid formed from the decomposition of organic matter such as plankton and algae that sank to the bottom of the ancient sea. The remains of this organic matter are gradually covered by sediment, and over millions of years, they transform into crude oil. Crude oil is a mixture of thousands of different chemical substances, most of which are hydrocarbons, that is, molecules in which carbon and hydrogen atoms are bonded in various arrangements. The molecules contained in crude oil range from simple ones like propane and butane to complex ones like asphaltenes.
Crude oil varies in its composition of hydrocarbons and other molecules depending on the region of origin. For example, crude oil produced in Canada's oil sands contains many heavy molecules, while crude oil from Saudi Arabia's Ghawar oil field contains many light molecules. Crude oil extracted from the Brent oil field in the North Sea has a low sulfur content, while crude oil extracted from the Gulf of Mexico has a high sulfur content.
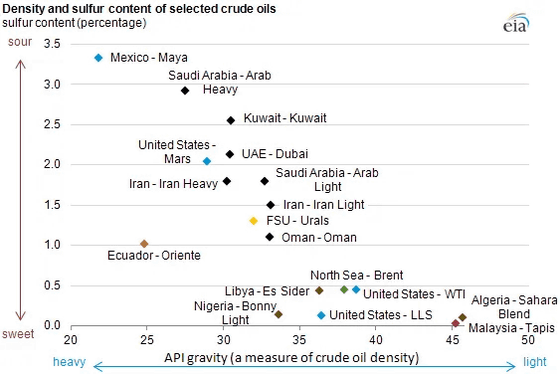
The role of an oil refinery is to properly process this mixture of hydrocarbons and other molecules. In other words, it separates the mixture into individual chemicals or groups of chemicals and uses various chemical reactions to convert lower-value chemicals into higher-value, more useful chemicals.
Oil refineries employ various methods to separate and process crude oil. Of these, distillation is the most important process. The various molecules in crude oil boil at different temperatures and condense into a liquid at different temperatures. Basically, small, light molecules boil and condense at lower temperatures, while large, heavy molecules boil and condense at higher temperatures. This boiling point range can be represented by a distillation curve.
The following is a distillation curve, showing which parts of the crude oil boil at what temperature. In this example, half of the crude oil boils at 350 degrees Celsius, and approximately 80% boils at 525 degrees Celsius. Since the molecules contained in different types of crude oil vary, the distillation curve will also differ.
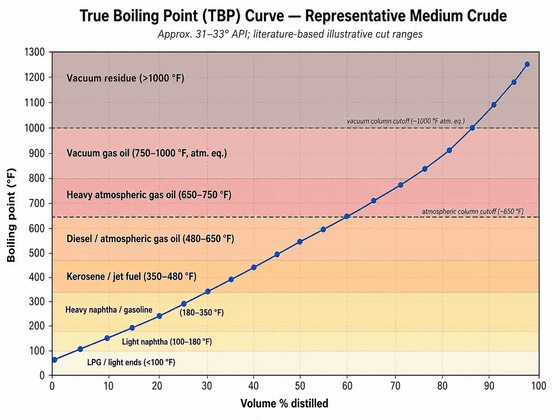
Substances obtained from crude oil are often 'mixtures of chemical substances' defined by their boiling point ranges. For example, gasoline is not a single chemical substance, but rather a mixture of hydrocarbons consisting mainly of molecules with 4 to 12 carbon atoms. The U.S. Energy Information Administration defines gasoline as having a boiling point range of 122 to 158 degrees Fahrenheit at a 10% distillation temperature and 365 to 374 degrees Fahrenheit at a 90%
In oil refineries, this range of boiling and condensation points is used to separate crude oil into petroleum and other chemicals through distillation. In the distillation process, salts are first removed, and then the mixture is heated to 650-750 degrees Fahrenheit, turning most of it into vapor. This vapor is sent to trays (filled with liquid) at different heights, where it cools and condenses into liquid as it rises. Heavier molecules with the highest boiling points condense in the lowest tray, while lighter molecules condense in the highest tray. This allows for the separation of different molecules.
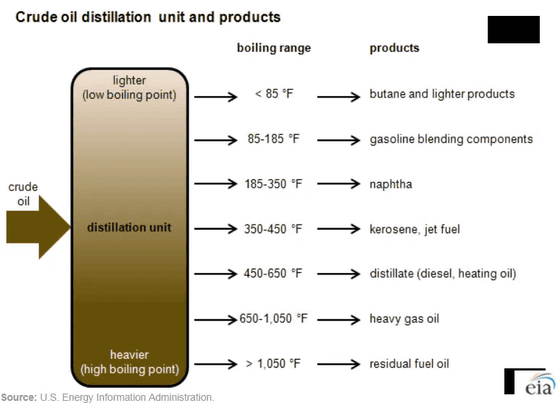
In oil refineries, crude oil is distilled into various fractions in distillation columns, but the types of fractions separated vary from refinery to refinery. In simple oil refineries, the process may end with just this first distillation step, 'atmospheric distillation.' However, most oil refineries go through further processes to further process the fractions.
The gas that comes out of a distillation column performing atmospheric distillation is a mixture of several different light molecules, such as propane, methane, butane, and isobutane. To separate this mixture into its constituent gases, the oil refinery sends the gas to a gas plant. The gas plant has multiple distillation columns designed to condense various substances from the mixture, first separating butane, propane, and other light gases, and then separating butane and propane. As a result, lighter gases come out of the top of the distillation column, and heavier liquids come out of the bottom. The heavier molecules that remain without evaporating during distillation are called residues and are not of much value.

Next, in oil refineries,
The heavy fraction obtained by atmospheric distillation is mixed with a catalyst, and heat and pressure are applied to break down the heavy molecules into lighter molecules. The catalyst is then separated from the mixture using a cyclone separator. In other words, by rotating the mixture, the heavier catalyst is separated from the rest of the mixture, washed, and reused. The cracked oil is then sent to another distillation column where it is separated into various fractions.
In addition to cracking, various processes are used in oil refineries to alter the chemical structure of different molecules. Catalytic reforming involves exposing naphtha fractions to heat and pressure in the presence of a catalyst to produce a mixture of new chemicals called reformed oil, which is used in gasoline refining. Isomerization processes alter the physical arrangement of various molecules, such as butane, to produce isomers that have the same chemical formula but different structural arrangements. Hydrogenation involves reacting various crude oil fractions with hydrogen in the presence of a catalyst to remove impurities and improve quality.
To store the various raw materials and products generated during these processes, oil refineries have a vast number of storage tanks called tank farms. These storage tanks can hold millions of gallons of liquid. Gases such as propane and butane are usually stored as pressurized liquids in above-ground tanks, underground cavities, or salt domes.

Potter said of oil refining, 'The oil refining process itself is often surprisingly simple, at least conceptually. What surprises me is the sheer scale of it. Oil refining is an expensive business not necessarily because the process is complex, but because of the enormous amount of crude oil that has to be processed. For example, the oil refinery that Chevron built in Richmond is the size of a small city.'
Related Posts:
in Science, Posted by logu_ii







