'The poorness of the new coronavirus antibody test is a disaster level,' said the CEO of a major pharmaceutical company, and what is the problem with the antibody test?
Severin Schwan, CEO of Swiss-based global pharmaceutical company
'A disaster': Roche CEO's verdict on some COVID-19 antibody tests-Reuters
https://www.reuters.com/article/us-roche-results/a-disaster-roche-ceos-verdict-on-some-covid-19-antibody-tests-idUSKCN2240JS
Roche CEO Blasts Faulty Antibody Tests, Touts Own Product-Bloomberg
https://www.bloomberg.com/news/articles/2020-04-22/roche-ceo-blasts-faulty-virus-tests-while-touting-own-product
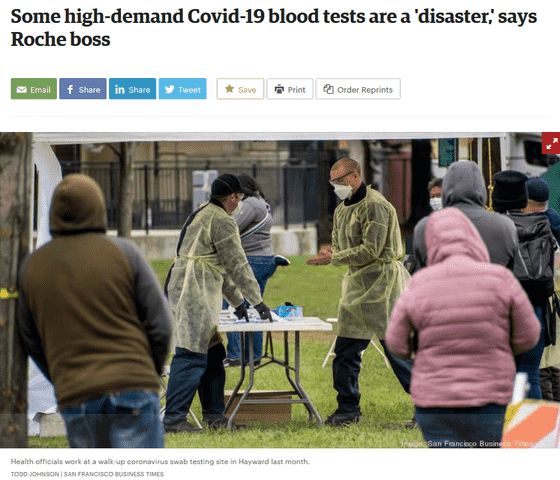
Some high-demand Covid-19 blood tests a 'disaster,' says Roche CEO Severin Schwan-San Francisco Business Times
https://www.bizjournals.com/sanfrancisco/news/2020/04/22/covid-coronavirus-serologic-blood-tests-roche.html
When infected with COVID-19,
In addition, antibody testing can be performed by a relatively simple method, such as puncturing a fingertip with a needle and collecting a blood sample. It also has the advantage that the time to obtain results is short and the test kit is cheap.
However, Roche's CEO Schwann said in a telephone press conference on April 22 that he announced results for the first quarter of 2020, `` We tested off-the-shelf antibody tests on the market, but the reliability of these test kits It wasn't very satisfying, it was a disaster, and the ethics of the companies that sell these test kits are very questionable, 'he said. did.

Schwan's severe criticism of ready-made antibody test kits is due to 'unreliability.' Mr. Schwan said, 'Any amateur can make an antibody test kit. If I and someone else spend the night, I can develop an antibody test kit in my garage. In other words, the problem is that something that doesn't work as a test kit is actually for sale. '
Schwan isn't the only one questioning the accuracy of antibody testing. Former Secretary-General of the US Food and Drug Administration (FDA) Scott Gottlieb said in a CNBC news show on May 4th: 'If you test positive for an antibody, we recommend that you test again. Frankly speaking, I will receive it three times, 'he said , pointing out the high false positive rate.
On the other hand, antibody tests with high accuracy have also appeared. On May 3, the FDA approved the antibody test ' Elecsys Anti-SARS-CoV-2 ' newly developed by Roche. According to Roche, this antibody test uses blood taken from the veins of the test subjects and detects both two types of antibodies, ' IgM ' and ' IgG ', so the probability of correctly determining negative subjects is high. Is 99.8%, and if positive, it is 100% and reliable. This antibody test drug has been sold to medical institutions and research institutions throughout Japan since May 13th.
Related Posts:
in Note, Posted by darkhorse_log