Study finds Indian generic drugs more likely to cause serious adverse events

EXPRESS: Are All Generic Drugs Created Equal? An Empirical Analysis of Generic Drug Manufacturing Location and Serious Drug Adverse Events - In Joon Noh, John Gray, George Ball, Zachery Wright, Hyunwoo Parks,
https://journals.sagepub.com/doi/10.1177/10591478251319691
All generic drugs are not equal, study finds
https://news.osu.edu/all-generic-drugs-are-not-equal-study-finds/
In Japan, generic drugs are only allowed to be sold if the quality, efficacy, and safety of the original drugs are confirmed to be equivalent by the Pharmaceuticals and Medical Devices Agency (PMDA) . The US Food and Drug Administration (FDA) has similar regulations, but the research team pointed out that 'the pharmaceutical supply chain is opaque, and the impact of pharmaceutical manufacturing in emerging countries such as India on quality risks has not been verified.' To answer this question, the research team collected information on 2,443 types of pharmaceuticals and investigated the relationship between country of origin and safety.
The study found that generic drugs made in India had a 54.3% higher risk of causing serious adverse events, including death, than those made in the US. George Ball of the Indiana University Kelley School of Business, a member of the research team, said, 'Generic drugs can vary greatly depending on where they are manufactured. The FDA claims that all generic drugs are as safe as brand-name drugs, but this is not the case for generic drugs made in India.'
John Gray of Fisher College, a member of the research team, commented, 'Indian-made generic drugs, especially those that have become more susceptible to price pressure over time since they were on the market, have a significantly higher incidence of serious adverse events than American-made generic drugs.' 'Current generic drug price competition is based on the premise that 'all generic drugs are created equal.' We hope that the results of this study will encourage a broader discussion of generic drug price competition.' Gray also called for two improvements from the FDA: 'End the practice of announcing inspection schedules for overseas factories in advance' and 'Improve transparency of manufacturing locations and quality.'
John Gray on LinkedIn: EXPRESS: Are All Generic Drugs Created Equal? An Empirical Analysis of… |
https://www.linkedin.com/posts/john-gray-4a83544_express-are-all-generic-drugs-created-equal-activity-7294056999351115777-yEli

In Japan, 172 generic drug manufacturers conducted voluntary inspections and reported on November 18, 2024 that 3,796 items, or 43.5% of the items inspected, had 'differences between the approval document and the actual manufacturing process.' However, the differences were limited to 'inputting small amounts in small portions in a process where the approval document stated that the entire amount was to be put in at once' and 'misprinting of 'bi' and 'pi',' and no serious cases requiring voluntary recalls or other measures were confirmed.
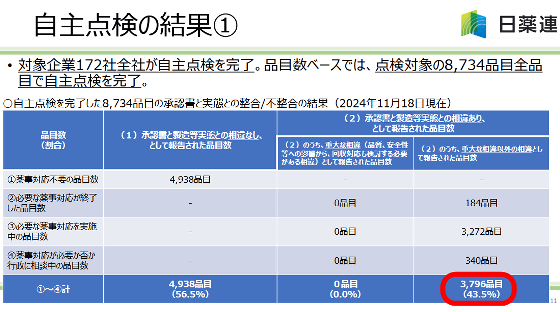
The report of the inspection results by the Federation of Pharmaceutical Manufacturers' Associations of Japan can be found at the link below.
Results of voluntary inspections regarding consistency between manufacturing and sales approvals for generic drugs and the actual manufacturing and testing methods
https://www.mhlw.go.jp/content/10807000/001333723.pdf

Related Posts:
in Science, Posted by log1o_hf