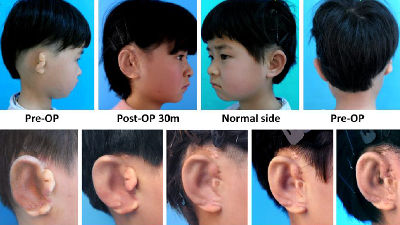
A vaccine to treat skin cancer has started Phase I clinical trials, and possibility of application to other cancers as well

Health, Labor and Welfare issuedAnnual trend of death / mortality rate by cause of deathAccording to 1981 to 2009, the most common cause of death in Japan is malignant neoplasia (cancer), and furtherDemographics surveyIt is also known that more than 30% of the cause of death in 2012 is cancer. A new cancer vaccine has entered the phase of Phase I clinical trial (examination of the safety of vaccine for humans for human) in order to make such cancer troubles complete.
Cancer vaccine begins Phase I clinical trials | Harvard School of Engineering and Applied Sciences
https://www.seas.harvard.edu/news/2013/09/cancer-vaccine-begins-phase-i-clinical-trials
A new cancer vaccine research project,Harvard School of Engineering and Applied SciencesMr. David J. Mooney, who served as a professor at Wyss Institute and also a core member of the Wyss Institute,Dana-Farbergun InstituteAt the cancer vaccine center leader,Harvard Medical SchoolThe research team of Glenn Dranoff and his colleagues, who is also a member of the professor and the Wyss Institute, is proceeding.
A vaccine effective for cancer treatment so far is to first remove immune cells from the body of a patient,ReprogrammingAnd then return it to the patient's body. In this project, instead of the previous method,FDAAuthorized bypolymerUse a sponge shaped like a fingernail disk (disc) made from fingers.

This sponge is injected under the skin,Patient's immune cells are intensified and reprogrammed in the bodyIt is designed so that immune cells go around the body and attack cancer cells. This eliminates the need to remove immune cells from the body.
The Science magazineScience Translational MedicineIn the preclinical stage posted in the clinical stage, 50% of the mice were treated perfectly with two doses of vaccine and the other half of the mice died within 25 days in melanoma. Also, this technique is a kind of skin cancerMelanomaAlthough it was designed as a vaccine, the possibility of application to other cancers is also ant.

ByNovartis AG
"Our vaccine has been made possible by combining extensive biomedical expertise," says Mooney, one of the team leaders. Also, "This is a collaborative research model of Wyss InstituteBridge researchIt will be the first treatment to reach human clinical trials. In addition, this bridging research approach is useful to advance technology engineering to the clinical stage much faster than traditional academic environments, "says Don Ingber, founder of the Wyss Institute.
The Phase I clinical trial of this cancer vaccine is expected to be completed in 2015, and here seems to be done to evaluate whether it is a safe vaccine even if it is put into the human body. As Dr. Dranoff, one of the project leaders, said, "Not only is it very rare for new technologies to reach these clinical trials in humans as soon as possible", not only as a mere skin cancer vaccine but also a wide specialty Combining knowledge, you may be getting a lot of attention as a style of research projects that can quickly develop new treatments.
Related Posts:
in Science, Posted by logu_ii