Seven diabetics died due to an undisclosed bug in a blood glucose monitor, and some say that life-threatening medical devices should be made public.
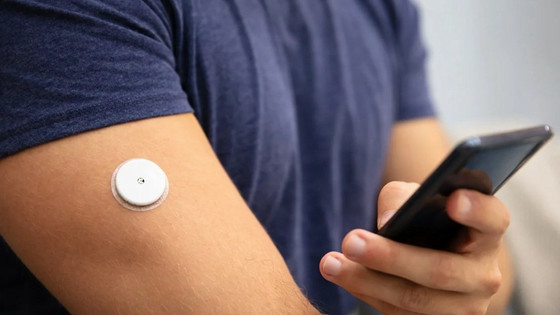
Medical device manufacturer Abbott sells the Freestyle Libre Plus, a continuous glucose monitor (CGM) worn by diabetics, in the United States. A malfunction with the Freestyle Libre Plus has been reported to the FDA (Food and Drug Administration) resulting in over 700 injuries and seven deaths.
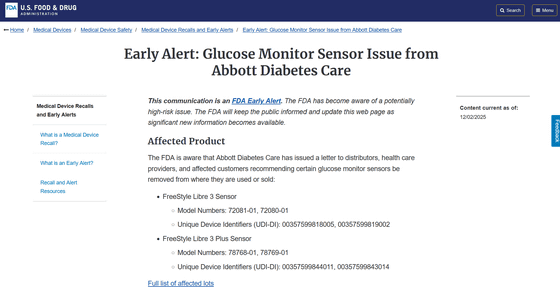
Early Alert: Glucose Monitor Sensor Issue from Abbott Diabetes Care | FDA
https://www.fda.gov/medical-devices/medical-device-recalls-and-early-alerts/early-alert-glucose-monitor-sensor-issue-abbott-diabetes-care
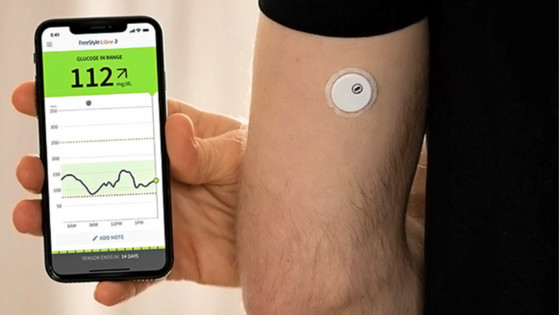
This defect causes the device to display an extremely low blood sugar level even when the blood sugar level is not actually low. Patients with advanced diabetes believe this display and ingest sugar to raise their blood sugar level, but because they ingest sugar when their blood sugar level is not actually low, this can cause serious health problems and be life-threatening.
Bradley M. Kuhn of the Software Freedom Conservancy (SFC) wrote a blog post about the issue, claiming that he himself was diagnosed with diabetes in 2025 and used the company's products.
Seven Diabetes Patients Die Due to Undisclosed Bug in Abbott's Continuous Glucose Monitors - Conservancy Blog - Software Freedom Conservancy
https://sfconservancy.org/blog/2025/dec/23/seven-abbott-freestyle-libre-cgm-patients-dead/
Kuhn is particularly concerned about the proprietary nature of medical device software and designs. He argues that if the product's technical specifications and source code were publicly available, they could be peer-reviewed by engineers around the world, improving safety and reliability. He points out that, at present, manufacturers' disclosure of information is extremely vague, making it unclear whether the cause of a malfunction is hardware or software, raising serious concerns from a public health perspective.
Kuhn is concerned that most users of Abbott's app agree to the company's liability disclaimer, which could hinder legal action such as class action lawsuits. In fact, it's estimated that less than 1% of patients use the app without agreeing to its terms.

To combat this situation, Kuhn is exploring technological approaches. Specifically, he is seeking collaborators to disassemble and reverse-engineer used devices, extracting software from them and analyzing how the hardware works, in order to create a safe environment unconstrained by manufacturer restrictions. He also supports the development of Juggluco, an open-source blood glucose management app, with the aim of releasing it for anyone to use.
Furthermore, Kuhn pointed out the contradiction between the lot numbers of products subject to FDA recall and those listed as safe on Abbott's verification website, saying, 'How can we trust software from a company that can't even accurately verify information with life-saving medical devices?' and called for transparency.
Related Posts:







