Why doesn't the Earth's inner core, which is hotter than the Sun, melt?

By
It is known that the surface temperature of the inner core of the center of the earth, which is 96% composed of iron as a solid, is higher than the surface temperature of the sun. The reason why the inner core can keep a solid without melting even though it is hotter than the sun has been a long-standing mystery for scientists, but a paper that may put an end to that mystery has been published. I did.
Stabilization of body-centred cubic iron under inner-core conditions: Nature Geoscience: Nature Research
http://www.nature.com/ngeo/journal/vaop/ncurrent/full/ngeo2892.html
New theory explains how Earth's inner core remains solid despite extreme heat | KTH
https://www.kth.se/en/aktuellt/nyheter/new-theory-explains-how-earth-s-inner-core-remains-solid-despite-extreme-heat-1.705398
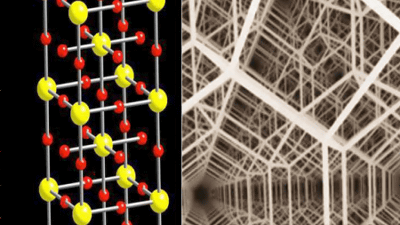
The inner core closest to the center of the earth is a crystal such as iron, which is about the size of the moon. The atomic-level crystal structure of the inner core, which is unimaginably large, changes its shape depending on the ambient temperature and pressure, like all metals. Under normal temperatures and atmospheric pressure, iron has a ' body-centered cubic lattice structure (BCC)' in which atoms are located at the vertices and centers of each cubic unit cell, but extremely high pressure is applied. It changes to a ' hexagonal close-packed structure (HCP)' in which atoms are located inside the hexagonal prism at each corner of the top and bottom of the regular hexagonal prism.
The inner core of the earth is under pressure about 3.5 million times that of the earth's surface, and the temperature is over 6000 degrees Celsius. In this environment, it was proposed that the atomic structure of iron in the inner core might change from BCC to HCP, but in a paper published in February 2017 by a research team at the Royal Institute of Technology of Sweden. , It was announced that the atomic structure of iron in the inner core retains BCC. According to Dr. Anatoly Veronoschko, who led the study, a computational model of iron larger than the iron treated in previous studies was investigated, and iron with a BCC structure was not found in the past in the inner core environment. It is said that it showed an atomic diffusion pattern.

By
In the survey, a simulation was conducted with Triolith, which is considered to be one of the largest supercomputers in Sweden. As a result, it was found that the atomic BCC becomes unstable at low temperatures and stabilized at high temperatures. Normally, the diffusion of an atom destroys its crystal structure and transforms it from a solid to a liquid. However, although the atomic structure diffuses in the environment of the Earth's inner core, it seems that the structure of BCC can be maintained in such a way that other atoms move to a certain place. It sounds a little difficult, but you can understand it by watching a movie that explains this phenomenon in an easy-to-understand manner.
How Earth's inner core remains solid despite heat --YouTube
At extremely high temperatures and pressures, atoms begin to diffuse and become unstable. When an atom becomes unstable, it cannot maintain its crystal structure and becomes a liquid.

However, in the extreme environment of the inner core, the crystal structure is maintained by replacing other atoms, although the atomic structure becomes unstable, as if the playing cards were shuffled. As a result, the iron in the inner core of the earth is kept as a solid.

Related Posts: