Why has a cure for Alzheimer's disease still not been found, even after more than 100 years of research?

Despite Alzheimer's disease being officially documented in 1906 and being studied for over 100 years, and despite the National Institutes of Health (NIH) investing approximately $4 billion (about 637.9 billion yen) annually in Alzheimer's and dementia research, a cure to stop or reverse cognitive decline has yet to be found. The economics podcast Freakonomics Radio delves into why developing an effective cure has been so difficult, based on interviews with Charles Piller, an investigative journalist for the scientific journal Science, and Matthew Schlag, a neurologist at Vanderbilt University Medical Center.
Why Has There Been So Little Progress on Alzheimer's Disease? - Freakonomics

◆Even after more than 100 years of research, there is still no cure.
Alzheimer's disease can progress in the brain long before symptoms such as memory impairment appear, and according to Freakonomics Radio, changes in the brain may begin as early as 20 years before symptoms appear. In the United States, more than 7 million people are affected by Alzheimer's disease, and in addition to memory impairment, problems with cognitive function, physiological function, and behavior appear.
Alzheimer's disease was officially recorded as a disease in 1906. German physician Alois Alzheimer examined the brains of women who had suffered from memory loss and hallucinations after their deaths and discovered that their brains were shrunken, that there were tangles of nerve fibers, and that there were strange deposits.
Since then, researchers have continued to investigate what these 'deposits' or 'tangles' are and how they relate to Alzheimer's disease. The NIH's funding for Alzheimer's disease and dementia research has increased from approximately $1 billion (approximately 159.5 billion yen) per year to approximately $4 billion (approximately 637.9 billion yen) per year over the past decade, making it the second largest research funding after cancer.
However, Pillar points out that while progress has been made in developing treatments for cancer, diabetes, and heart disease, no treatment has yet been found in Alzheimer's disease research that can stop or reverse the decline in cognitive function.

◆The amyloid cascade hypothesis was at the heart of the research.
In considering the stagnation in the development of these treatments, Freakonomics Radio is focusing on the hypotheses that have underpinned Alzheimer's disease research over the years.
According to Schlag, Alzheimer's disease involves the accumulation of protein clumps in the brain. Of particular importance are amyloid-beta , which clumps on the outside of brain cells, and tau protein, which clumps inside nerve cells. It is believed that when tau protein clumps abnormally, the function of nerve cells is impaired, leading to brain atrophy and memory impairment.
Since around 1990, amyloid-beta has been at the center of Alzheimer's disease research. The hypothesis that clumps of amyloid-beta act as the 'first domino' of the disease, followed by a complex chain reaction that leads to the death of nerve cells, is called the 'amyloid cascade hypothesis,' and research and drug development have progressed in accordance with this hypothesis.
Schlag points out the problems with the amyloid cascade hypothesis, stating, 'Firstly, this idea may not be correct, and secondly, biology is likely to be much more complex.' However, Schlag does not believe that amyloid-beta is not involved in Alzheimer's disease, explaining that 'treating only amyloid-beta as the cause of the disease may not be sufficient to control Alzheimer's disease as a whole.'

◆Suspicions arise regarding image data in important research paper
The amyloid cascade hypothesis was once significantly shaken. A clinical trial to remove amyloid-beta with a vaccine failed, leading researchers to question whether amyloid-beta was truly the main cause of Alzheimer's disease. Subsequently, the explanation that it was not the large amyloid plaques, but rather smaller amyloid-beta clumps called 'oligomers' that were negatively affecting nerve cells, gained attention.
In this context, one of the important studies supporting the amyloid cascade hypothesis that attracted attention was the paper ' A specific beta-amyloid protein assembly in the brain impairs memory ,' published in Nature in 2006. The research team that wrote this paper reported that when they extracted the protein from the brains of mice genetically modified to overproduce amyloid protein and injected it into the brains of rats, the rats showed signs of cognitive decline.
Sylvain Lesnet, co-author of the paper, argued that clumps of a specific type of amyloid-beta oligomer are the main cause of Alzheimer's disease. Pillar states that this research brought renewed attention to the amyloid cascade hypothesis in the field of Alzheimer's disease research and became one of the most cited experiments in the history of Alzheimer's disease research by 2022.
However, Schlag later came to believe that there were problems with the images used as evidence in this paper. The images of ' Western blotting, ' which examines the types and amounts of proteins, were particularly problematic. Western blotting analyzes proteins by looking at the intensity of lines and stains in the image, so changes in the image's intensity affect the interpretation of the experimental results.
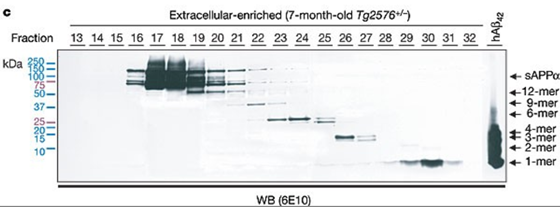
The problems that Schlag discovered were then confirmed by Pillar with other image analysis experts and Alzheimer's disease research specialists. As a result, Pillar stated that 'the images appeared to have been manipulated to support hypotheses that were not supported by the actual data.'
This 2006 Nature paper was ultimately retracted at the request of co-author Karen Ash. In a separate memo, Ash acknowledged that there were problems with the data, but explained that she 'was unaware of the problems until they were pointed out.'
A key paper on the causes of Alzheimer's disease may contain fabricated images - GIGAZINE
◆ Criticism of the system for investigating research misconduct
After doubts arose about the paper, the image data wasn't the only issue. Pillar believes there are also problems with the system for investigating suspected research misconduct and how it is handled.
The NIH acknowledged the potential problems with the 2006 Nature paper and referred the matter to the Office of Research Integrity (OLI) of the U.S. Department of Health and Human Services. However, the OLI is a small organization and does not have the personnel to investigate all research misconduct allegations on its own. Therefore, the investigation was ultimately returned to the University of Minnesota, where the research at the center of the allegations was conducted.
Pillar criticizes the direction of this investigation. By entrusting the investigation to the university where the allegations are made, the university is forced to examine issues that affect its own research and reputation. Pillar points out that 'the university itself has the greatest loss from conducting a thorough and open investigation.' Details of the University of Minnesota's investigation have not been made public, but it is reported that the university has asked academic journals to consider retracting other papers by Resne.

◆Image issues also found in papers by influential figures in research funding.
Pillar focused not only on individual papers but also on papers by researchers who were in positions to influence the direction of Alzheimer's disease research. One person Pillar cited as particularly important was Eliezer Masria, who headed the neuroscience division at the National Institute on Aging in the United States.
Masuria was in a position to oversee an annual research budget of approximately $2.7 billion, and was a figure who could have a significant influence on the direction of neuroscience research. Pillar, along with Schlag, Mu Yang of Columbia University, independent researcher Kevin Patrick, and scientific image analysis expert Elizabeth Bick, examined Masuria's paper.
As a result, 132 of the papers examined contained instances where images appeared unnatural or inappropriately reused. Specifically, some images had unnatural parts, some appeared to have been manipulated, some appeared to have been reused inappropriately, and some appeared to have been used in different papers to support different claims. Pillar and his colleagues did not examine all 800 of Masuria's papers, but they compiled the concerns they found into a 300-page document.
Pillar sent documents to Masria requesting a rebuttal to the allegations, but Masria did not respond. The NIH subsequently announced that Masria had left his position.
Eliza Masria, a renowned neuroscientist and head of a department at the National Institute on Aging, is suspected of image falsification in a paper widely cited worldwide on Alzheimer's and Parkinson's disease - GIGAZINE

◆How are drugs that target amyloid being evaluated?
Research based on the amyloid cascade hypothesis has led to the development and approval of actual drugs. As an example, Pillar cites Aduhelm, an antibody drug that targets amyloid-beta. Aduhelm was approved by the FDA, but was later withdrawn from the market.
Pillar has harshly criticized Aduhelm, calling it 'dangerous and ineffective,' and has also raised concerns about the close relationship between the FDA, which reviews drugs, and the pharmaceutical company that develops them, regarding Aduhelm's approval. According to Pillar, Billy Dunn of the FDA, who was involved in the approval of Aduhelm, was found to have worked closely with the company seeking approval.
Researchers criticize 'Alzheimer's drug' approved by regulatory authorities, stating it carries a very high risk of brain swelling - GIGAZINE
Pillar is not claiming that antibody drugs targeting amyloid-beta are completely ineffective, stating that there is 'evidence that FDA-approved antibody drugs can slow cognitive decline very slightly.' However, he explains that the effect may be so small that clinicians, patients, and families do not notice it, and that the patient's symptoms themselves do not improve.
◆Where will future research be headed?
These criticisms do not mean that amyloid-beta is unrelated to Alzheimer's disease. Schlag argues that Alzheimer's disease and neurodegenerative diseases should be re-evaluated as 'diseases in which waste products accumulate in the brain and the brain is unable to properly eliminate them.' Amyloid-beta plaques and tau protein entanglements are also part of these waste products, and attention should be paid not only to amyloid-beta but also to the pathways through which waste products are removed from the brain.
Schlag argues that focusing on vascular health and blood pressure management, which are part of the pathways that remove waste products from the brain, may provide clues to preventing and treating Alzheimer's disease.
Furthermore, Pillar is not rejecting science itself, but rather states that universities, academic journals, the NIH, and the FDA—organizations involved in evaluating, funding, and approving research results—should be more careful and responsible in order to prevent scientific knowledge from being distorted by false data or corruption.
Related Posts:
in Science, Posted by log1b_ok