Clinical trial results show that the oral drug 'orfogliprone' for treating obesity and diabetes can improve blood sugar and weight more than oral semaglutide

Efficacy and safety of once-daily oral orforglipron compared with oral semaglutide in adults with type 2 diabetes (ACHIEVE-3): a multinational, multicentre, non-inferiority, open-label, randomized, phase 3 trial - The Lancet
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(26)00202-3/abstract
expert reaction to ACHIEVE-3 trial results about efficacy and safety of once-daily oral orforglipron compared with oral semaglutide in adults with type 2 diabetes | Science Media Center
https://www.sciencemediacentre.org/expert-reaction-to-achieve-3-trial-results-about-efficacy-and-safety-of-once-daily-oral-orforglipron-compared-with-oral-semaglutide-in-adults-with-type-2-diabetes/
A research team led by Julio Rosenstock, an endocrinologist at the University of Texas Southwestern Medical Center, and Daisuke Yabe of the Kyoto University Graduate School of Medicine, published the results of a Phase III trial comparing the oral GLP-1 drug orfoglipron, manufactured by Eli Lilly, with oral semaglutide tablets in adult type 2 diabetes patients with inadequate blood sugar control while being treated with metformin, one of the first medications prescribed to those diagnosed with type 2 diabetes. Phase III trials are conducted to verify efficacy and safety in a large number of patients under conditions similar to actual treatment.
The 52-week trial involved 131 sites in Argentina, China, Japan, Mexico, and the U.S. The researchers divided 1,698 participants into four groups:
- Group administered 12 mg of orfogliprone
- Group administered 36 mg of orfogliprone
- Group receiving oral semaglutide 7 mg
- Group receiving oral semaglutide 14 mg
The research team focused on the reduction of hemoglobin A1c (HbA1c) , an index that reflects average blood sugar levels over the past one to two months, as the main endpoint. The lower this number, the better the blood sugar status.

The average HbA1c at the start of the study was 8.3%. The research team reported the following changes in HbA1c at 52 weeks:
Orfoglipron 12 mg group: -1.71%
Orfoglipron 36 mg group: -1.91%
Oral semaglutide 7mg group: -1.23%
Oral semaglutide 14mg group: -1.47%
The research team confirmed that the study achieved its primary endpoint of non-inferiority, which indicates that the new drug (orfogliprone) is not inferior to the existing drug (oral semaglutide) in clinical trials. Furthermore, the study concluded that orfogliprone demonstrated not only non-inferiority but also superiority, demonstrating a greater improvement in HbA1c than oral semaglutide tablet.
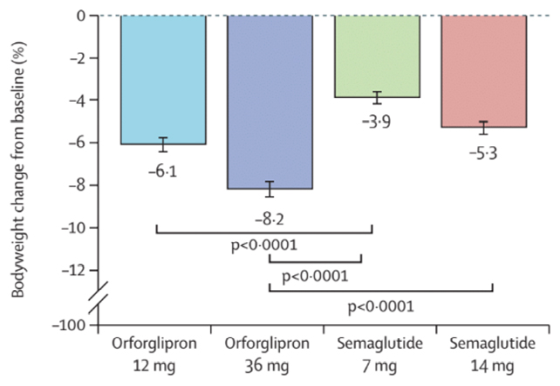
The orfogliprone group also showed greater weight loss. The weight loss rates reported by the research team are as follows:
Orfoglipron 12 mg group: -6.1%
Orfoglipron 36 mg group: -8.2%
Oral semaglutide 7mg group: -3.9%
Oral semaglutide 14mg group: -5.3%
The research team explains that orfogliprone is designed to be taken daily without food or water restrictions. This difference may be beneficial for daily treatment, as complex dosing conditions can lead to forgetting or inconsistent dosing.
However, there are some concerns about side effects, as the research team noted that orfogliprone caused more gastrointestinal symptoms, such as nausea and diarrhea, and many patients discontinued treatment due to these side effects. Orfogliprone also showed a greater increase in mean heart rate.
In response to the results of this study, Marie Spreckley of the University of Cambridge praised it as 'a large-scale, direct comparison and clinically important,' but pointed out that 'the high number of discontinuations due to side effects is an issue that may affect the ease of continuation in actual clinical practice,' and '(This study) was conducted for one year, so longer-term safety and cardiovascular outcomes remain a challenge.' Cardiovascular outcomes are indicators that show the results of treatments or interventions in research on heart and vascular diseases.
The research team concluded that by directly comparing oral GLP-1 drugs under the same conditions, it became easier to evaluate not only their effectiveness but also practical differences, such as whether side effects would allow treatment to continue.
Related Posts:
in Science, Posted by log1b_ok